2 M cells mainly depend on l K r and not on l K s and therein lies their propensity for pronounced APD prolongation when conditions favour it, especially in cases where depolarizing currents ( I Na, I Ca – late sodium current has also been found inherently more potent in these cells, thus constituting a second mechanism accounting for the more prolonged baseline APD) 6,7 increase. The latter can be categorised as rapid (l K r) and slow (l K s) delayed currents, mediated by the HERG (new nomenclature Kv11.1) and KCNQ1 (new nomenclature Kv7.1) channels, respectively. More specifically, those generated by the sustained opening of long (L)-type calcium channels (then triggering sarcoplasmic reticulum calcium release through ryanodine-sensitive calcium channels leading to excitation – contraction coupling), as well as a late sodium current (through the Nav1.5 channel also responsible for phase 0) on one hand and potassium currents on the other. Regarding the ionic substrate, the plateau of action potential (phase 2) is due to antagonism between positive inward and outward ( Figure 2) currents. 4 Of crucial importance, APD in these cells is more susceptible to changes leading to its prolongation as compared to cells in the other myocardial layers. They may also potentially affect APD of neighbouring endo/epicardial cells though gap junction coupling. Thus, they form a layer, or area, more susceptible to causing unidirectional block (larger possibility for an extrasystole to fall into the absolute refractory period), all the more so if baseline APD prolongation exists. 3 However, a defined population of cells, mainly located in the intermediate layers of myocardium (M cells), but also found in islets nearer the epi- and endocardium, 4,5 exhibits a more protracted action potential because of their unique ion channel properties, as explained later on. The normal sequence of repolarization 1,2 begins at the epicardial layer, moving (not propagating, except for the small contribution of gap junctions) towards the endocardium, based on the intrinsic action potential duration (APD) of the respective cells. bundle/fascicle block) are prone to cause increases in the overall QT interval, not only by delaying cycle initiation for certain areas but also through a direct effect on ion movement (by affecting stretch-activated channels). 
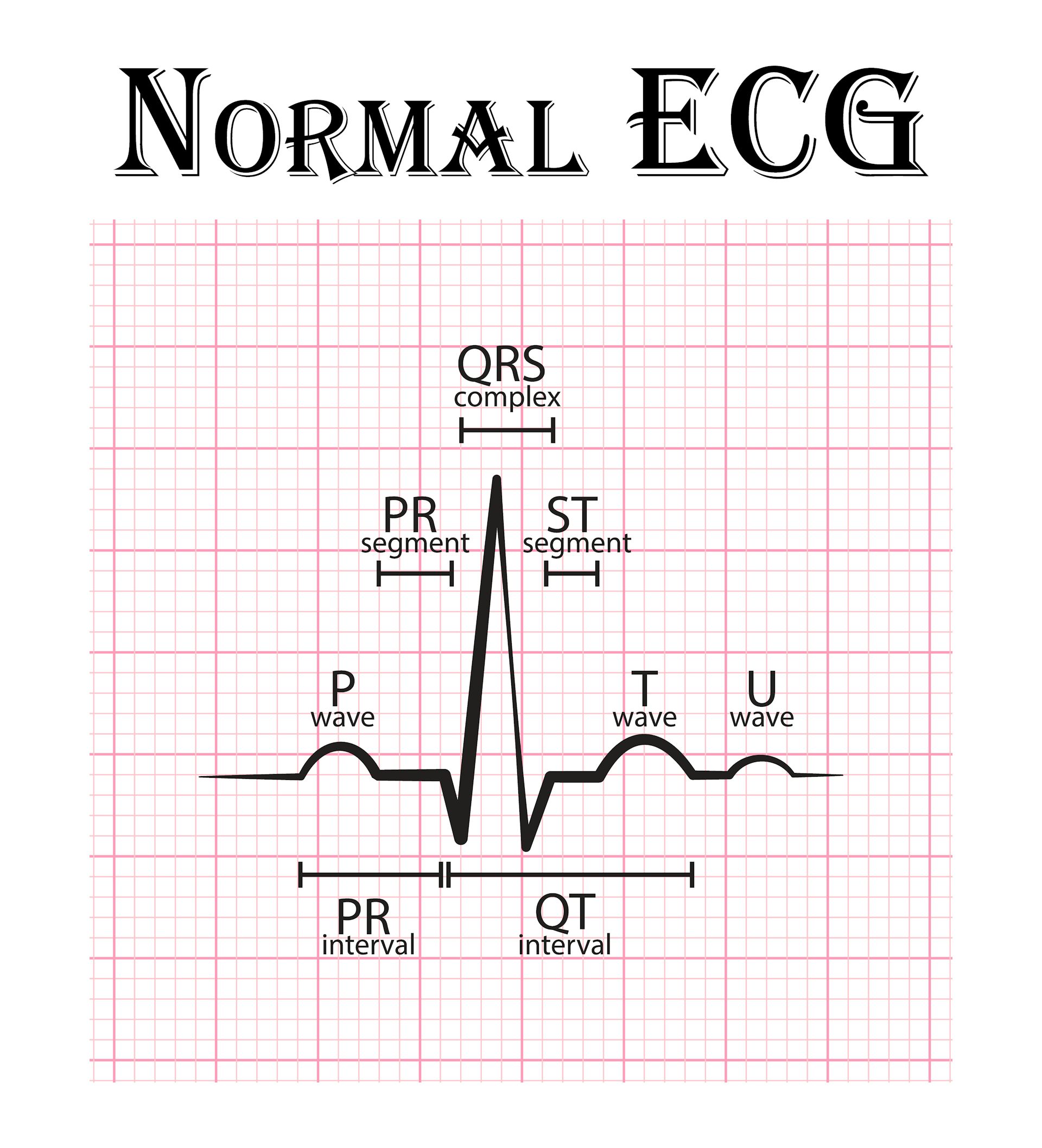
It follows that even changes in the velocity of depolarization propagation (i.e. The QT interval is a more or less easily identifiable segment in the ECG and thought to visualise the full cycle of ventricular depolarization and repolarization. The TQ interval corresponds to phase 4 of the action potential. 
Thus, the QRS complex reflects the summed depolarization (phase 0), the ST segment the transient current equilibrium leading to a brief electrical quiescence (phase 2) and the T wave the summed repolarization (phase 3) of the action potential ( Figure 1). The surface ECG represents a time and space integral to the individual cells’ action potentials, with the ‘time’ parameter also incorporating the delay in activation between myocardial areas, as opposed to solely the time course of the phenomenon in each cell. (Patho)Physiology of QT Prolongation and Related Arrhythmias In the following short review, focused on QT prolongation, it will be attempted to summarise the underlying (patho)physiology of this entity and the extent to which it constitutes a problem with regard to both prevention and management of malignant, potentially lethal arrhythmic events in various substrates. More specifically, disorders may involve abnormal initial repolarization rate (phase 1 of the action potential – Brugada syndrome) or abnormally shortened/prolonged late repolarization (phase 3 – short and long QT syndromes, respectively). 
Given that different segments and layers of the myocardium have divergent baseline properties and consequently respond differently to stimuli, it is not surprising that several arrhythmic disorders of the myocardium, both congenital and acquired, are related to derangements in precisely this repolarization process. Ventricular repolarization, as opposed to depolarization, is not a triggered phenomenon following an orderly sequence, hence the dissimilarity between their inscribed electrocardiographic waves rather, ventricular myocytes repolarize at a time and rate determined by their intrinsic electrophysiological properties (relative concentration of ion channel types and isoforms), as well as by the preceding electrical and mechanical events that affect the former. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
